Phosphorus tribromide Basic informationProduct Name:Phosphorus tribromideSynonyms:PHOSPHORUS TRIBROMIDE;PHOSPHORUS(III) BROMIDE;PHOSPHORIC (III) BROMIDE;PHOSPHORUS TRIBROMIDE, 1.0M SOLUTION IN DICHLOROMETHANE;PHOSPHORUS TRIBROMIDE SOLUTION APPROX.;PHOSPHORUSTRIBROMIDE APPROX. 1 MOL/L IN&;PHOSPHORUS TRIBROMIDE, 99.99+%;PHOSPHOROUS TRIBROMIDE (5263)CAS:7789-60-8MF:Br3PMW:270.69EINECS:232-178-2Product Categories:Thiophenes;BrominationMicro/Nanoelectronics;C-X Bond Formation (Halogen);Electronic Chemicals;Others;Synthetic Reagents;Inorganics;metal halide;Aliphatics;Intermediates & Fine Chemicals;Pharmaceuticals;1Mol File:7789-60-8.mol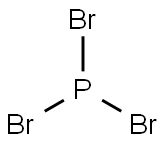 Phosphorus tribromide Chemical PropertiesMelting point -41.5 °C (lit.)Boiling point 175 °C (lit.)density 2.88 g/mL at 20 °C (lit.)vapor density 9.3 (vs air)vapor pressure 0.27 psi ( 54 °C)refractive index n20/D 1.697(lit.)Fp 172.9°Cstorage temp. Store below +30°C.solubility soluble in Ether,Acetone,Chloroformform LiquidSpecific Gravity2.88color Clear to slightly hazy colorless to yellow or light brownOdorIrritating odourWater Solubility reactsFreezingPoint -40℃Sensitive Moisture SensitiveMerck 14,7357BRN 3903060Stability:Stable, but reacts violently with water. Incompatible with moisture, strong bases, strong oxidizing agents, reactive metals, acids, alcohols.InChIKeyIPNPIHIZVLFAFP-UHFFFAOYSA-NLogP2.394 (est)CAS DataBase Reference7789-60-8(CAS DataBase Reference)NIST Chemistry ReferencePhosphorus tribromide(7789-60-8)EPA Substance Registry SystemPhosphorus tribromide (7789-60-8)Safety InformationHazard Codes CRisk Statements 34-37-40-14-67Safety Statements 26-45-36/37-36/37/39RIDADR UN 3264 8/PG 2WGK Germany 2RTECS TH4460000F 21TSCA YesHS Code 2812 90 00HazardClass 8PackingGroup IIHazardous Substances Data7789-60-8(Hazardous Substances Data)MSDS InformationProviderLanguageACROSEnglishSigmaAldrichEnglishALFAEnglishPhosphorus tribromide Usage And SynthesisChemical PropertiesPhosphorus tribromide is a colourless liquid(possibly hazy) with the formula PBr3. It fumes in air due to hydrolysis and has a penetrating odour. It is widely used in the laboratory for the conversion of alcohols toalkyl bromides. Phosphorus tribromide Chemical PropertiesMelting point -41.5 °C (lit.)Boiling point 175 °C (lit.)density 2.88 g/mL at 20 °C (lit.)vapor density 9.3 (vs air)vapor pressure 0.27 psi ( 54 °C)refractive index n20/D 1.697(lit.)Fp 172.9°Cstorage temp. Store below +30°C.solubility soluble in Ether,Acetone,Chloroformform LiquidSpecific Gravity2.88color Clear to slightly hazy colorless to yellow or light brownOdorIrritating odourWater Solubility reactsFreezingPoint -40℃Sensitive Moisture SensitiveMerck 14,7357BRN 3903060Stability:Stable, but reacts violently with water. Incompatible with moisture, strong bases, strong oxidizing agents, reactive metals, acids, alcohols.InChIKeyIPNPIHIZVLFAFP-UHFFFAOYSA-NLogP2.394 (est)CAS DataBase Reference7789-60-8(CAS DataBase Reference)NIST Chemistry ReferencePhosphorus tribromide(7789-60-8)EPA Substance Registry SystemPhosphorus tribromide (7789-60-8)Safety InformationHazard Codes CRisk Statements 34-37-40-14-67Safety Statements 26-45-36/37-36/37/39RIDADR UN 3264 8/PG 2WGK Germany 2RTECS TH4460000F 21TSCA YesHS Code 2812 90 00HazardClass 8PackingGroup IIHazardous Substances Data7789-60-8(Hazardous Substances Data)MSDS InformationProviderLanguageACROSEnglishSigmaAldrichEnglishALFAEnglishPhosphorus tribromide Usage And SynthesisChemical PropertiesPhosphorus tribromide is a colourless liquid(possibly hazy) with the formula PBr3. It fumes in air due to hydrolysis and has a penetrating odour. It is widely used in the laboratory for the conversion of alcohols toalkyl bromides.Phosphorus tribromide (PBr3) is commonly used in Hell-Volhard-Zelinsky halogenation for the α-bromination of carboxylic acids to form the corresponding acyl bromide. It is also useful for the conversion of primary and secondary alcohols to alkyl bromides.UsesPhosphorus(III) bromide is used mainly for the conversion of alcohols to alkyl bromides and it is used in tests for skin corrosivity. It is used for alkene preparation and it act as a lewis base and a lewis acid. It is used as a catalyst for the alpha-bromination of carboxylic acids and used as intermediates in hell-volhard-zelinsky halogenation. Phosphorus tribromide is used in the manufacture of pharmaceuticals such as alprazolam, methohexital and fenoprofen. It is also used as a fire suppression agent.UsesPhosphorus tribromide is used mainly for the conversion of alcohols to alkyl bromides and it is used in tests for skin corrosivity. It is used for alkene preparation and it act as a lewis base and a lewis acid. Phosphorus tribromide is used in the manufacture of pharmaceuticals such as alprazolam, methohexital and fenoprofen. Phosphorus tribromide may be used as a reagent: In a novel protocol for synthesizing 7-ethoxy-1-(p-ethylphenoxy)-3,7-dimethyl-2-octene, a synthetic juvenile hormone mimic of trans,trans,cis-10,11-epoxy-7-ethyl-3,11-dimethyltrideca-2,6-dienoate. To synthesize 1,2,3-diazaphosphinane, 1,2,3-thiazaphosphinine and 1,2-azaphosphole bearing a chromone ring. To synthesize 1-Bromoundec-1-ene from 10-undecen-1-ol.PreparationPhosphorus tribromide, PBr3, is most conveniently prepared by reaction between liquid bromine and a solution of white phosphorus in PBr3. In most of its reactions, the tribromide resembles the trichloride although the former has been much less studied and in some cases the products seem to be more complex.ReactionsSilyl phosphites such as (EtO)2POSiMe3 and (Et3SiO)3P are known. The latter compound can be prepared in 30% yield by reacting phosphorus tribromide with an organosilane in the presence of zinc chloride. Esters of the former type can be made by reaction. 3Et3SiO+PBr3–-(Et3SiO)3+3RBr Et3SiONa+(EtO)2PCl–-(EtO)2POSiEt3+NaCl)General DescriptionA colorless fuming liquid with a pungent odor. Corrosive to metals and tissue. Boiling point 347°F (175°C). Freezing point -40°F (-40°C).Air & Water ReactionsFumes in air. Decomposed by water to form phosphoric acid and hydrobromic acid. Reaction with warm water is very rapid and may be violent [Mellor v.8. 1032 1940].Reactivity ProfilePhosphorus tribromide reacts with oxidizing agents to generate heat and products that may be flammable, combustible, or otherwise reactive; the reactions may be violent. Forms complexes with potassium or sodium metal that explode when shocked. Drop wise addition to 3-phenylpropanol caused an explosion when stirring of the mixture was discontinued [Chem. Brit., 1974, 10, 101-102].Health HazardInhalation causes severe irritation of nose, throat, and lungs. Ingestion causes burns of mouth and stomach. Contact with eyes or skin causes severe burns.Safety ProfileProbably highly toxic. A corrosive irritant to the eyes, skin, and mucous membranes. Wdl react with water, steam, or acids to produce heat, toxic and corrosive fumes. Violent reaction or ignition with calcium hydroxide + sodium carbonate, phenylpropanol, sulfuric acid, oleum, fluorosulfuric acid, chlorosulfuric acid, 1,1,1 -tris(hydroxymethyl)methane, water, potassium, sodium, RuO4. When heated to decomposition it emits very toxic fumes of Brand POx. See also PHOSPHIDES and BROMIDES.Purification MethodsIt is decomposed by moisture, it should be kept dry and is corrosive. Purify it by distillation through an efficient fractionating column [see Whitmore & Lux J Am Chem Soc 54 3451] in a slow stream of dry N2, i.e. under strictly dry conditions. [Gay & Maxson Inorg Synth II 147 1946, Org Synth Col Vol II 358 1943.] Dissolve it in CCl4, dry it over CaCl2, filter and distil it. Store it in sealed ampoules under N2 and keep it away from light. HARMFUL VAPOURS.Phosphorus tribromide Preparation Products And Raw materialsRaw materialsGovernment regulation–>PhosphorusPreparation Products2-(1,5-Dimethyl-1H-pyrazol-3-yl)acetonitrile ,97%–>1-(2-ETHOXYETHYL)PIPERAZINE–>5-ACETAMIDO-2-BROMOPYRIDINE–>2-METHOXY-5-(2′-ETHYLHEXYLOXY)BENZENE-1&–>2-BROMO-4-(TRIFLUOROMETHYL)PYRIMIDINE–>3,3,3-Trifluoropropionic acid–>4-BROMO-6-(TRIFLUOROMETHYL)PYRIMIDINE–>4-TERT-BUTYLCYCLOHEXANECARBOXYLIC ACID–>2-HYDROXY-5-NITROBENZYL BROMIDE–>2-(4-CHLORO-PHENYL)-ETHANESULFONYL CHLORIDE–>3-(3-Hydroxyphenyl)-DL-alanine–>6-Fluoro-4-(trifluoromethyl)quinolin-2-amine ,97%–>4-(Trifluoromethyl)quinolin-2-amine–>4-TRIFLUOROMETHYL-QUINOLINE-2-CARBOXYLIC ACID–>3,5-DIMETHOXYPHENYLACETONITRILE–>4-(Trifluoromethyl)quinoline-2-carbohydrazide ,97%–>2-(2-CHLORO-PHENYL)-ETHANESULFONYL CHLORIDE–>(4-(Trifluoromethyl)quinolin-2-yl)methanol ,97%–>3-BROMO-CYCLOPENT-2-ENOL–>TETRAHYDRO-2H-PYRAN-4-YLACETYL CHLORIDE–>(6-Fluoro-4-(trifluoromethyl)quinolin-2-yl)methanol ,97%–>2-(4-FLUORO-PHENYL)-ETHANESULFONYL CHLORIDE–>Ethyl 6-fluoro-4-(trifluoromethyl)quinoline-2-carboxylate ,97%–>Cyclohexylamine hydrobromide–>2-Bromo-3-nitropyridine–>Tetrahydropyranyl-4-acetic acid–>2-(2-FLUORO-PHENYL)-ETHANESULFONYL CHLORIDE–>5-Bromomethyl-1,3-dimethyl-1H-pyrazole ,97%–>2-Bromo-6-fluoro-4-(trifluoromethyl)quinoline–>(2-FLUOROBENZYL)METHYLAMINE–>2-BROMO-4-(TRIFLUOROMETHYL)QUINOLINE–>(4-FLUORO-BENZYL)-METHYL-AMINE–>3,5-DIMETHOXYBENZYL BROMIDE–>1-(2-Methoxyethyl)hydrazine–>4-BROMO-TETRAHYDROPYRAN–>N-(2-BROMO-3-PYRIDINYL)ACETAMIDE–>Cyclohexylmethyl bromide–>2-Bromoethyl ethyl ether–>2-Fluoroethyl bromide |
| Tag:Phosphorus tribromide(7789-60-8) Related Product Information |