| Betulin Basic information |
| Extract Physical and Chemical Properties Extraction method Identification method Physiological functions Storage References |
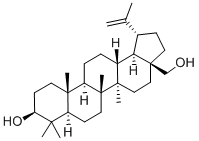
| Product Name: | Betulin |
| Synonyms: | Lup-20(29)-ene-3,28-diol, (3β)-;BETULIN(P);BETULIN hplc;Betulin,Betulinol,Betulol,Betulenol;3b,28-Dihydroxylup-20(29)-ene;Betulinic alcohol;SpirulinaPowder&Tablets;etulin |
| CAS: | 473-98-3 |
| MF: | C30H50O2 |
| MW: | 442.72 |
| EINECS: | 207-475-5 |
| Product Categories: | Triterpenoids;Tri-Terpenoids;natural product;Pentacyclic Triterpenes;chemical reagent;pharmaceutical intermediate;phytochemical;reference standards from Chinese medicinal herbs (TCM).;standardized herbal extract;473-98-3 |
| Mol File: | 473-98-3.mol |
 |
| Betulin Chemical Properties |
| Melting point | 256-257 °C(lit.) |
| alpha | D15 +20° (c = 2 in pyridine) |
| Boiling point | 493.26°C (rough estimate) |
| density | 0.9882 (rough estimate) |
| refractive index | 1.5045 (estimate) |
| storage temp. | 2-8°C |
| solubility | Chloroform (Slightly), Methanol (Slightly, Heated) |
| form | Off-white powder |
| pka | 15.10±0.10(Predicted) |
| color | Pale Brown to Beige |
| Merck | 14,1189 |
| Stability: | Hygroscopic |
| InChIKey | FVWJYYTZTCVBKE-ROUWMTJPSA-N |
| LogP | 8.607 (est) |
| CAS DataBase Reference | 473-98-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Betulin(473-98-3) |
| Safety Information |
| Hazard Codes | Xn,Xi |
| Risk Statements | 36/37/38-20/21/22-68/20/21/22-68 |
| Safety Statements | 36/37-36-26 |
| WGK Germany | 3 |
| RTECS | OK5755000 |
| HS Code | 29181985 |
| Provider | Language |
|---|---|
| Betulin | English |
| SigmaAldrich | English |
| Betulin Usage And Synthesis |
| Extract | Betulin is an extract from bark of the white birch tree, which has been known since the 18th century and is chemically defined. Betulin was discovered by German-Russian chemist Johann Tobias Lowitz. He was the first scientist to study and characterise betulin, and in doing was one of the first to isolate an active plant ingredient. Betulin is a substance of pure plant origin that gives the birch bark its typical white colour. Betulin protects birch trees from environmental effects, such as extreme temperatures, pest infestations and solar radiation. |
| Physical and Chemical Properties | It is white crystalline powder, soluble in alcohol, chloroform and benzene, slightly soluble in cold water, petroleum ether. |
| Extraction method | Currently Betulin is obtained mainly through direct extraction, and mainly by solvent reflux extraction and recrystallization purification Specifically: 1. Extract betulin from ethanol solution. After heating reflux about 5h, the extract is vacuum distillated and recrystallized with ethanol for 2 to 3 times with ethanol to obtain crude product of betulin. 2. The crude product was recrystallized from methanol/chloroform (1: 1) (every 80g crude product requires 300mL methanol/chloroform solution).The mixture is allowed to stand overnight and then suction filtered to obtain white needle-like betulin. 3. Ultrasonic treatment for 20min when refluxing is carried out, and the extraction yield and product purity of betulin can be improved through the destruction of bark organization. 4. The optimum conditions of supercritical CO2 fluid extraction are as follows: the amount of modifier was 115mL/(g birch bark powder); extraction pressure is 20MPa; extraction temperature was 55 ℃; liquid CO2 flow rate was 10kg/h. |
| Identification method | HPLC conditions for betulin: Isolated Constituent: Betulin(10μl sample injection) Column Stationary Phase: C18(150×2)mm 5μ Column Mobile phase: Acetonitrile/water =30:70 Flow rate (ml/min);0.2Detection Wavelength (nm):270 Column Temperature:30℃ |
| Physiological functions | Betulin and its derivatives as biological agents has shown great potential in the treatment of HIV and cancer by interfering with the post-life cycle of the virus, which is related to the entry as well as growth and maturing of virus. As an effective anti-tumor drug, it can directly cause certain types of tumor cells to start self-destruction of the apoptosis program, and can slow down the growth of several types of tumor cells.It can reduce dietary induced obesity, reduce lipid content in serum and tissue and improve insulin sensitivity.It has mild anti-inflammatory properties at higher concentrations, and its anti-inflammatory properties are largely due to inhibition of non-neural gene pathways.Betulin in the body can inhibit the maturation of sterol regulatory element binding protein (SBERPs), thereby reducing the biosynthesis of cholesterol and fatty acid.In addition, with its anti-inflammatory, anti-virus effect and functions of inhibiting protein dissolution in the hair fiber, improving the luster of damaged hair and promoting hair growth and other activities, it can be used in food, cosmetics and pharmaceutical industries. |
| Storage | Cool and dry, kept from light and temperature. |
| References | http://www.imlan.de/en/infothek/what-is-betulin.html |
| Description | Sterol regulatory element binding protein 2 (SREBP-2) regulates cholesterol synthesis by activating the transcription of genes for HMG-CoA reductase and other enzymes of the cholesterol synthetic pathway. When cellular sterol levels are high, SREBP is bound by SCAP and Insig to ER membranes as a glycosylated precursor protein. Upon cholesterol depletion, the protein is cleaved to its active form and translocated into the nucleus to stimulate transcription of genes involved in the uptake and synthesis of cholesterol. Betulin, the precursor of betulinic acid, is a pentacyclic triterpene found in the bark of birch trees. Betulin inhibits the SREBP-driven pathway of cholesterol and fatty acid biosynthesis by promoting SCAP–Insig binding which prevents the activation and release of SREBP-2 from the ER. At 15-30 mg/kg/day, betulin has been shown to decrease lipid levels and increase insulin sensitivity in mice fed a western-type diet. In an atherosclerosis disease model, 30 mg/kg/day betulin can reduce the size and improve the stability of atherosclerotic plaques in LDLR-knockout mice. At 2.5-5 μg/ml betulin, in combination with cholesterol, demonstrates anticancer effects by inducing apoptosis in Jurkat cells, A549 lung carcinoma cells, and HeLa cervical carcinoma cells. |
| Chemical Properties | crystals |
| Uses | Betulin may be used in the preparation of betulinic acid, which shows anti-HIV, antimalarial and anti-inflammatory activities. |
| Uses | birch bark extract is described as having anti-irritant and antiseptic properties, and effective in acne treatment. It is used to make sunburn products, soothing lotions, and aftershaves. The oil is astringent and is mainly used for its curative effects, especially in cases of acne and eczema. In folkloric medicine, birch bark extract was considered good for bathing skin eruptions. Destructive distillation of the bark’s white epidermis yields an empyreumatic oil known as oil of birch tar, Oleum rusci, or dagget. This is a thick, bituminous, brownish-black liquid with a pungent, balsamic odor. It contains a high percentage of methylsalicilate, creosol, and guaiacol and is almost identical to wintergreen oil. |
| Uses | antineoplastic, antihyperlipidemia |
| Definition | ChEBI: A pentacyclic triterpenoid that is lupane having a double bond at position 20(29) as well as 3beta-hydroxy and 28-hydroxymethyl substituents. |
| General Description | A cell-permeable pentacyclic triterpenoid from birch bark extract that interacts with SCAP (SREBP cleavage activating protein) and prevents SREBP (sterol regulatory element-binding protein) Golgi proteolytic activation (1 to 13.55 μM in Rat hepatocytes CRL-1601) via a similar mechanism as oxysterols by inducing the association of SCAP with Insig-1 (insulin-induced gene-1), thereby causing SCAP-SREBPs complex ER retention. Unlike oxysterols, Betulin does not activate LXR to induce concomitant HMGCR (HMG-CoA reductase) degradation and SREBP-1 up-regulation. While both Betulin and HMGCR inhibitor Lovastatin (Cat. No. 438185) inhibit cellular cholesterol synthesis (by ~70% in CRL-1601 cultures with respective compound at 13.55 and 1 μM concentration), only Betulin suppresses cellular fatty acid synthesis (by ~55% at 13.55 μM in CRL-1601). Betulin is shown to exhibit comparable in vivo efficacy as Lovastatin (both dosed at 30 mg/kg/day with chow) in ameliorate high fat/cholesterol diet-induced obesity (% fat/lean tissue ratio increase from non-fat diet mice = 167, 44, and 33 in mice consuming fat diet alone, with Lovastatin, with Betulin, respectively). However, only Betulin is demonstrated to lower fasting blood glucose and insulin levels and reduce fat cell size in white adipose tissue in high fat diet mice. Fatostatin (Cat. No. 341329) in comparison does not target SCAP via sterol-binding site, nor does it induce SCAP binding to Insig-1. |
| Anticancer Research | In addition, triterpenoids (betulin and its derivatives) isolated from HSSEactivated the signaling pathway regulated by p53 family genes, leading to the inhibition of BC cell viability or even the induction of apoptosis. And also, theseresearchers found that all the triterpenoids had no effect on normal breast cells.These findings provide an important basis for the use of those triterpenoids in thedevelopment of alternative therapies for breast cancer treatment (Hsu et al. 2015). |
| Betulin Preparation Products And Raw materials |
| Raw materials | Betulin-3-acetate–>BETULIN DIACETATE–>Betulinic acid |
| Preparation Products | betulonic aldehyde |