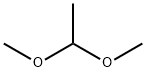
| Description | 1,1-Dimethoxyethane is an aliphatic hydrocarbon which is found in figs, leek, and black tea. It gives a sharp ethereal, fruity and green note. It is used as a flavoring agent in food. 1,1-Dimethoxyethane is also used as a reagent for the diol protection and condensation reactions. |
| Preparation | To a flask equipped with a mechanical stirrer, condenser, and gas addition tube and containing 10 gm of a 6 3% solution of boron trifluoride in methanol is added 1.0 gm of mercuric oxide and 200 gm (6.25 moles) of methanol. Then 70 gm (3.13 moles) of acetylene is added with vigorous stirring at room temperature. After the reaction the catalyst is neutralized with aqueous potassium carbonate, the product is extracted into ether, dried, and distilled to afford 104 gm (37%), b.p. 64°-65°C.
 |
| Reference | Evaluation of Certain Food Additives and Contaminants: Fifty-seventh report of the Joint FAO/WHO Expert Committee on Food Additives, 2001, ISBN 92-4-120909-7
M. J. Taschner, Encyclopedia of Reagents for Organic Synthesis, 2001, ISBN 9780471936237 |
| Chemical Properties | 1,1-Dimethoxybenzene has a sharp, sweet, alcohol, ether, green, new-mown-hay odor |
| Chemical Properties | clear colorless liquid |
| Occurrence | Reported found in raspberry, blackberry, strawberry, peas, fresh figs, hop oil, coffee, cognac, white wine and tea. |
| Uses | Acetaldehyde dimethyl acetal may be used in the preparation of glucoside derivatives of steganol. It may be used as polymer solvent for the encapsulation of water-soluble model protein, bovine serum albumin into biodegradable poly(D,L-lactic acid. |
| Uses | As Mering’s mixture which is 2 vol dimethylacetal and 1 vol chloroform. |
| Uses | 1,1-Dimethoxyethane is used as a reagent in the synthesis of tricyclic and tetracyclic 1,5-benzodiazepine derivatives as nevirapine analogues. |
| Preparation | From acetaldehyde and methanol. |
| Definition | ChEBI: 1,1-dimethoxyethane is an acetal that is dimethoxymethane substituted by a methyl group at position 1. It has a role as a flavouring agent and a plant metabolite. It is an acetal and a diether. It is functionally related to a dimethoxymethane. |
| General Description | A liquid with a sharp odor. Less dense than water. Flash point between 0-73°F. Mildly toxic by ingestion and inhalation. Severely irritates the skin and eyes. Used to make other chemicals. |
| Air & Water Reactions | Highly flammable. May form unstable peroxides when exposed to oxygen. These products can sometimes be observed as clear crystals deposited on containers or along the surface of the liquid. Slightly soluble in water. |
| Reactivity Profile | 1,1-Dimethoxyethane may react violently with strong oxidizing agents. Can act as a weak base to form salts with strong acids and addition complexes with Lewis acids. In other reactions, which typically involve the breaking of the carbon-oxygen bond, ethers are relatively inert. |
| Health Hazard | Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control may cause pollution. |
| Fire Hazard | HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water. |
| Safety Profile | Mildly toxic by inhalation, ingestion, and skin contact. A skin and eye irritant. A very dangerous fire hazard when exposed to heat, flame, or oxiduzers. When exposed to heat or flame it can react vigorously with oxidizing materials. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also GLYCOL ETHERS. |
| Purification Methods | Distil the dimethyl acetal through a fractionating column and fraction boiling at 63.8o/751mm is collected. It forms an azeotrope with MeOH. Alternatively purify it as for acetal above. It has been purified by GLC. [Beilstein 1 IV 3103.] |