| Description | Dacomitinib is a selective and irreversible inhibitor of EGFR. It is a drug candidate for the treatment of non-small cell lung carcinoma. It is current under the Phase III clinical trials. It also shows potential for the treatment of HER-2 amplified breast cancer cells lines that are resistant to trastuzumab and lapatinib. |
| Biological activity | Dacomitibib (Dacomitinib, PF299804) is an effective and irreversible pan-ErbB inhibitor that is most effective in EGFR, with an IC50 of 6 nM. It is also highly effective in NSCLCs that carries EGFR or ERBB2 mutants (against Gefitinib) and EGFR T790M mutant. Phase 2.
Dacomitinib is taken orally once-daily. It is an irreversible inhibitor of HER-1 (EGFR), HER-2 and HER-4 tyrosine kinases. Dacomitinib targets multiple receptors of the HER pathway, whereas currently marketed HER-1 (EGFR) inhibitors for non-small cell lung cancer (NSCLC) target only one receptor in this pathway,developed by Pfizer. |
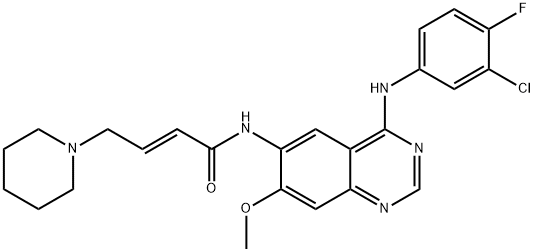
| Uses | Dacomitinib (PF299804), which name is (E)-N-[4-(3-chloro-4-fluoroanilino)-7- methoxyquinazolin-6-yl]-4-piperidin-1-ylbut-2-enamide, is a potent, orally available, highly selective, irreversible small-molecule tyrosine kinase inhibitor (TKI) of tyrosine kinase human epidermal growth factor receptors (HER) 1 (EGFR), HER2, and HER4. It is most effective against EGFR with IC50 of 6 nM. It is highly effective against NSCLCs carrying EGFR or ERBB2 mutant (anti-Gefitinib) and EGFR T790M mutant. |
| Clinical Study | Dacomitinib has advanced to several Phase III clinical trials. The results of the first trials were disappointing, with a failure to meet the study goals, Additional Phase III trials are ongoing.
Clinical evaluation of dacomitinib is ongoing in a number of clinical trials in patients with advanced NSCLC across lines of therapies and a range of histologies and molecular subtypes, such as EGFR and KRAS status.
Additionally, there is an ongoing clinical trial evaluating dacomitinib in recurrent and/or metastatic (RM) squamous cell carcinoma of the head and neck (SCCHN). |
| Description | The epidermal growth factor receptors (EGFR), whose members include ErbB1 (HER1), ErbB2 (HER2), ErbB3 (HER3), and ErbB4 (HER4), are receptor tyrosine kinases that are often over-expressed in cancer. PF-299804 is a second generation pan-ErbB receptor tyrosine kinase inhibitor (IC50s = 6, 45.7, and 73.7 nM for ErbB1, ErbB2, and ErbB4, respectively) that irreversibly binds to the ATP site in the catalytic domains of ErbB receptors. At 15 mg/kg, it demonstrates antitumor activity ranging from delay in tumor growth to complete regression in various tumor xenograft models expressing either wild-type ErbB or mutant ErbB family members that show resistance to first generation ErbB kinase inhibitors. |
| Uses | Dacomitinib is an Erb-inhibitor, or an inhibitor of the members of the epidermal growth factor receptor family. Used as an ant-cancer agent due to observation of over-expressed epidermal growth factor receptor in nearly all cases of squamous cell carcinoma in the head and neck. |
| Uses | PF299804 is a potent, irreversible pan-ErbB inhibitor against ErbB1, ErbB2 and ErbB4 with IC50 of 6 nM, 45.7 nM and 73.7 nM, respectively. |
| Definition | ChEBI: A member of the class of quinazolines that is 7-methoxyquinazoline-4,6-diamine in which the amino group at position 4 is substituted by a 3-chloro-4-fluorophenyl group and the amino group at position 6 is substituted by an (E)-4-(piperidin 1-yl)but-2-enoyl group. |
| in vitro | dacomitinib reduced the phosphorylation of her2, egfr, her4, akt, anderkin the majority of sensitive lines. dacomitinib exerted its antiproliferative effect through a combined g0–g1 arrest and an induction of apoptosis. dacomitinib inhibited growth in several her2-amplified lines with de novo and acquired resistance to trastuzumab. dacomitinib maintained a high activity in lines with acquired resistance to lapatinib. this study identifies her2-amplified breast cancer lines as most sensitive to the antiproliferative effect of dacomitinib and provides a strong rationale for its clinical testing in her2-amplified breast cancers resistant to trastuzumab and lapatinib [1]. |
| in vivo | to evaluate the in vivo efficacy of pf00299804, the authors generated xenografts in nu/nu mice using hcc827 gfp and hcc827 del/t790m cells and treated the mice with pf00299804. pf00299804 effectively inhibited the growth of hcc827 gfp xenografts. pf00299804 treatment was substantially more effective at inhibiting growth of this xenograft model than gefinitib. thus, these preclinical models suggest that pf00299804 may be quite effective against lung cancers that become resistant to gefitinib or erlotinib via acquisition of a t790m mutation in egfr [2]. |
| target | ErbB1 |
| IC 50 | 6 nm (egfr); 45.7 nm (erbb2); 73.7 nm (erbb4) |
| references | [1] kalous o, conklin d, desai aj, o’brien na, ginther c, anderson l, cohen dj, britten cd, taylor i, christensen jg, slamon dj, finn rs. dacomitinib (pf-00299804), an irreversible pan-her inhibitor, inhibits proliferation of her2-amplified breast cancer cell lines resistant to trastuzumab and lapatinib. mol cancer ther. 2012;11(9):1978-87.
[2] engelman ja, zejnullahu k, gale cm, lifshits e, gonzales aj, shimamura t, zhao f, vincent pw, naumov gn, bradner je, althaus iw, gandhi l, shapiro gi, nelson jm, heymach jv, meyerson m, wong kk, jfinne pa. pf00299804, an irreversible pan-erbb inhibitor, is effective in lung cancer models with egfr and erbb2 mutations that are resistant to gefitinib. cancer res. 2007;67(24):11924-32. |